Green Hydrogen vs Fossil Fuels: Comparing the Future of Energy
Author: Luke Wentlent, Ph.D., Principal R&D Engineer
The story is not a new one. Although fossil fuels built the world we know today, we now realize that they are also the element that could eventually take that world apart. You can see it in the searing heat of Phoenix, Arizona and the wildfires in Hawaii, Greece, and California. The arctic ice pack is melting. The planet’s temperature is rising. The first hurricane in more than 80 years just hit California. Climate change is real, and you can look to fossil fuels as its primary driver.
But do not lose hope! It’s time to act. While we all need to try to reduce our carbon footprint, the responsibly lies heavily on municipalities and corporations. The world is still largely fossil fuel dependent, and it will take great effort to move past them and build a new world. In this post, we’ll look at the two energy sources and where they stand today.
What is Green Hydrogen?
Green hydrogen is made using renewable energy, without greenhouse gas emissions. There are two technologies currently in use to produce green hydrogen at scale, alkaline water (ALK) electrolysis and proton exchange membrane (PEM) electrolysis. PEM electrolyzers are more expensive, but more efficient than ALK electrolyzers which have been in use longer.
Either way, green hydrogen is produced using electrolyzers to split water into hydrogen and oxygen. The hydrogen can be used, while the oxygen is vented harmlessly into the atmosphere. There is a significant opportunity to replace fossil fuels with green hydrogen. Hydrogen has the potential to decarbonize challenging industries such as steel production, last-mile delivery fleets, and aviation.
For many companies, the driving reason for developing green hydrogen is to reduce greenhouse gas emissions. Governments are increasingly putting incentives in places to make the switch. Public opinion also looks favorably on companies who act responsibly ahead of legislation which is likely to be next.
What are Fossil Fuels?
Fossil fuels are the fossilized remains of plants and animals that walked planet earth millions of years ago. Coal, oil, and natural gas are all fossil fuels. Because carbon is one of the major building blocks of life, these energy sources – when burned – are a primary source of carbon emissions.
Yet, for most of the 20th century, fossil fuels drove industry and created great wealth for the great nations of the world. Without free use of fossil fuels, the power and wealth of the world’s economies would be unthinkable. But our success comes at a cost for the earth itself.
Emissions are only part of the damage that burning fossil fuels does to the planet. Drilling, mining, and fracking for fossil fuels takes a tremendous toll on landscapes and ecosystems. In the quest for fossil fuels, entire forests are wiped out, mountaintops are removed, and groundwater is poisoned. In the long term – and increasingly in the present – fossil fuels are untenable if human life is to persist on the planet.
Comparing Green Hydrogen and Fossil Fuels
Fundamentally, hydrogen and fossil fuels are both energy carriers. To make use of that energy, we need to extract that energy from its carrier. For fossil fuels this typically means combusting the fuel with air in various types of engines or turbines. Hydrogen is more flexible. Energy can be extracted by either combustion or with a electrochemical means. To call either one “better,” we must define the context of what “better” means. That can be with respect to energy efficiency, GHG abatement, or cost, for example.
Fossil Fuels vs. Green Hydrogen: Energy Efficiency
Energy efficiency refers to the ultimate efficiency of the energy extraction process. How much of the energy stored in the carrier is used?
We know about fossil fuels. It is no exaggeration to say that they are a core pillar of our society. Fossil fuels powered the twentieth century and are still –nearly a quarter of a century in the twenty-first– our primary source for power generation today.
Given that there are a wide variety of ways in which combustion technologies are deployed, their ultimate energy efficiency will vary widely. Internal combustion engines have efficiencies between 20 and 40%, see figure below. Combined cycle gas turbines that run at power plants can be higher, into the 40% to mid-50% efficiency range.
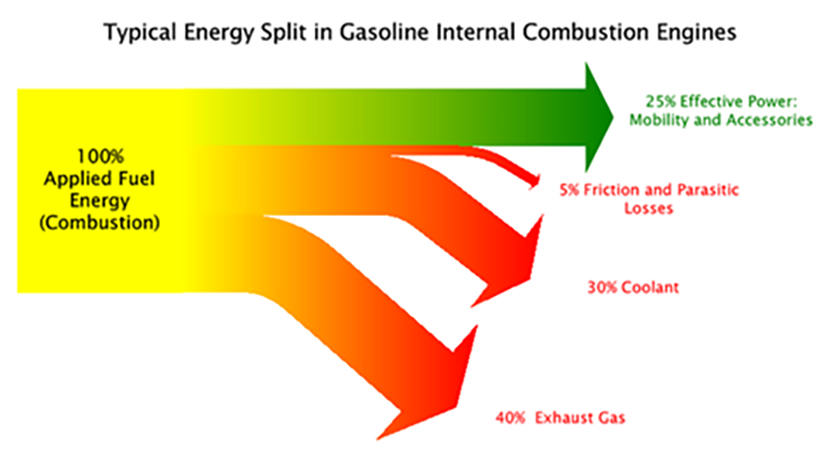
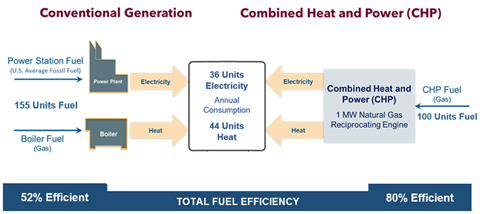
Combined heat and power plants are highly efficient combustion schemes where the excess heat generated is captured and employed, driving the efficiency up into the 65%-85% range. This is depicted in the chart below.
Combusting hydrogen achieves relatively similar efficiencies to fossil fuels. Hydrogen does have some combustion characteristics that afford it a more flexible operation than gasoline or natural gas; however, the ranges stated would be reasonable for a hydrogen combustion scheme in those applications.
Hydrogen can also be deployed in a fuel cell. These electrochemical devices which combine hydrogen gas with oxygen to produce a current can operate with efficiencies in the 50% – low-60% range, significantly better than most combustion technologies.
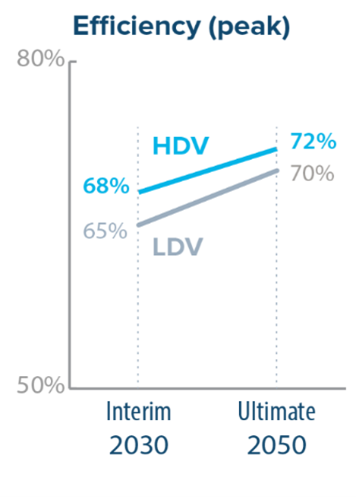
There are many fuel cell technologies (I.e., alkaline, PEM, molten carbonate). PEM, alkaline, and solid oxide achieve the best efficiencies, all of which are generally around 60%. Below are the DOE targets for fuel cell efficiency in light duty vehicles (LDV) and heavy-duty vehicles (HDV) in 2030 and 2050.
Fossil Fuels vs. Hydrogen: Environmental Impact
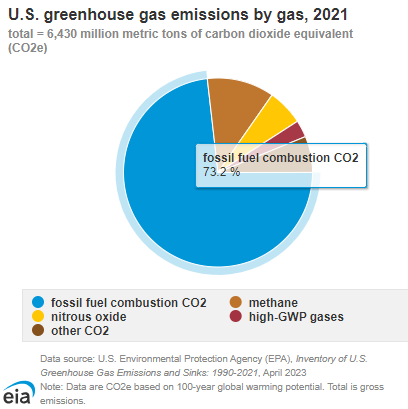
Another critical aspect to understand when discussing fossil fuels and hydrogen technologies is the environmental impact. This can mean many different things and be assessed in numerous ways. Let’s look at greenhouse gas emissions (GHG) and particulate matter (PM). According to the EPA, in 2021 fossil fuel combustion accounted for 73% of total GHG emissions and about 92% of total manmade CO2 emissions in the United States. We can point to numerous studies that show a clear, causal link between the increase in man-made GHG emissions and global warming.
The byproducts of combustion of fossil fuel depend, to a degree, upon the specific fuel and how it is burned. Typical byproducts include the following:
- Carbon Monoxide (CO)
- Carbon Dioxide (CO2)
- Sulfur
- Nitrogen Dioxide (NO2)
- Nitric Oxide (N2O)
- Volatile Organic Compounds (VOCs)
- Hydrocarbons (HCs)
Consider that burning one gallon of gasoline emits ~8.8 kg of CO2.
This is in stark contrast to hydrogen. Combusting hydrogen (H2) with air will only emit NOx — and there are methods to capture and minimize that. However, the true star is the hydrogen fuel cell, where the only byproduct of operation is water vapor. This chemically pure water can be put down the drain, released outside, or even reused to help clean the floors at some facilities.
Green Hydrogen Safety
Not only is green hydrogen sustainable, but hydrogen safety standards are also developed, mature, and effective, according to the Department of Energy (DOE).
Experts and regulators agree that when handled appropriately, hydrogen is a far safer fuel than those currently in use today, according to Washington State University professor Dr. Jacob Leachman. In fact, the U.S. Department of Energy’s Hydrogen and Fuel Cells Technologies Office stated “a number of hydrogen’s properties make it safer to handle and use than the fuels commonly used today,” including gasoline, natural gas, uranium, jet fuel, and diesel.
Aside from green hydrogen being produced solely from sustainable energy and only emitting harmless water as a by-product when used in a fuel cell, there are an abundance of hydrogen safety features which ensure production, distribution, storage, and use are as safe as possible.
The safety regulations, summarized here by the Fuel Cell & Hydrogen Energy Administration, are managed by governments, and regulated in accordance with third-party international hydrogen safety standards – making hydrogen a decarbonization solution with a safe, developed technical foundation. These include codes and standards around hydrogen infrastructure, on-road and fueling, first responder practices, fuel cell design, use in marine and aviation applications, and even for use in a fuel cell-powered bicycle.
Read our blog post:
Truth is Power: Hydrogen Safety 101
Green Hydrogen Scalability
A common point of discussion is the scalability of hydrogen and whether it can truly be matured to the point where it can make institutional change.
Hydrogen is not something new. It is the most abundant element in the universe and the world already uses it widely as a commodity. The United States alone annually produces ~10 million metric tons of hydrogen from steam methane reforming. In short, the world already knows how to handle, move, and store hydrogen.
However, the production of green hydrogen (electrolytic hydrogen powered by renewable resources) is admittedly nascent. Many green hydrogen production plants of this type have been proposed but few have broken ground. But things seem to be changing. The industry is currently at a tipping point where state and federal incentives are positioned to jump-start this needed scale up, akin to the battery and PV industry ramp up that have already occurred during the past 10-15 years.
The DOE is projecting that more than two million metric tons of green hydrogen will be required by the early 2030s. In order to meet these targets, we’ll need a massive scale up of the hydrogen production economy. Recognizing this, Congress passed a number of incentives and tax credits in the Inflation Reduction Act focused around catalyzing that acceleration along with all the supporting components (e.g., incentives for renewable development).
Conclusion
The world is entering into an unprecedented period where our energy systems will begin to radically change as new technologies become more widely available and enable things like green hydrogen to make significant inroads into our decarbonization targets. Virtually every climate scientist agrees that we must act now. We do not have the luxury of decades to delay addressing the global energy challenges we face.